Introduction:
The CLL2-BCG trial is a prospective, open-label, multicenter phase-II study based on the "sequential triple-T" (tailored, targeted, total eradication of CLL) concept proposed earlier [Hallek M., Blood 2013; 122(23): 3723-34]. This concept consists of sequentially applied combinations of targeted agents and aims for achieving undetectable minimal residual disease (MRD). It uses a sequential application of bendamustine (Ben) for debulking, followed by obinutuzumab (Obi) plus idelalisib (Ide) as induction and maintenance therapy for an all-comer population of physically fit and unfit, treatment-naïve (t-n) and relapsed/refractory (r/r) CLL patients (pts) irrespective of high-risk genetic markers.
Methods:
Pts with an absolute lymphocyte count ≥ 25.000/µl and/or lymph nodes ≥ 5 cm were to receive 2 cycles of Ben as debulking (70 mg/m² d1&2 q28 d), unless contraindicated. In the induction phase Obi 1000 mg was administered on d 1, 8 and 15 of cycle 1 and d1 of cycles 2-6; Ide was added in cycle 2 (150 mg twice daily). In the maintenance phase, daily dosing of Ide was continued and Obi was administered every 3 months until achieving a MRD-negative complete response or for up to 24 months. The primary endpoint was the overall response rate (ORR) at the end of induction therapy, secondary endpoints included MRD assessment, safety and survival.
Due to an increased incidence of opportunistic infections in other Ide trials, amendment 2 in March 2016 limited the recruitment to r/r CLL pts with high-risk features such as presence of a deletion 17p/TP53 mutation and/or ineligibility for ibrutinib treatment (refractoriness, intolerance or contraindications). Slow enrolment led to recruitment stop in September 2019.
Results:
Between May 2015 and September 2019, 48 pts were enrolled. Sixteen pts were t-n and 32 had r/r CLL with a median of 2 prior lines (range: 1-10); most common were BR and FCR, 6 pts each had received ibrutinib and venetoclax containing therapies. Median age was 66 (range 41-83) years, median CIRS score was 2 (0-13). Twenty-three pts (48%) were defined unfit by a CIRS score >6 (7 pts) and/or an impaired renal function with a Creatinine Clearance <70ml/Min (19 pts); 39 pts were male (81%). Nineteen pts (40%) had a del(17p) and/or TP53 mutation; 33 (70%) an unmutated IGHV status and 15 pts (42%) a complex karyotype, 36 pts (80%) had a high or very high CLL-IPI.
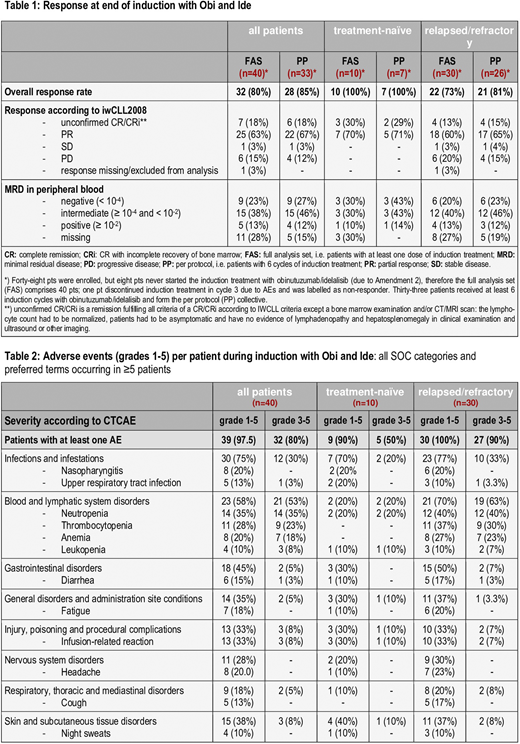
Thirty-eight patients (79%, 16 t-n and 22r/r) received Ben debulking. However, 8 pts never started the induction phase due to protocol amendment 2. Forty pts (10 t-n, 30 r/r) received induction treatment (FAS [full analysis set]), 33 completed the full 6 cycles (PP [per protocol] collective). Twenty-seven (7 t-n, 20 r/r) continued in a maintenance phase.
At the end of induction, 32 of 40 pts (FAS) and 28 of 33 pts with 6 induction cycles (PP) responded (ORR 80% and 85%, respectively); undetectable MRD levels (<10-4) by 4-color flow were achieved in 9 pts (23% and 27%, respectively) [Table 1]. Median progression-free survival was 44 months in t-n and 33 months in r/r CLL pts. Median overall survival was not reached for the t-n and 46 months in r/r pts; nine pts died, seven due to infections (two sepsis, including one in the context of severe enterocolitis, one pneumocystis jirovecii pneumonia and one influenza pneumonia, the other three after disease progression/start of subsequent treatment were considered unrelated to stud treatment), one cardiac arrest and one due to Richter´s transformation.
As of June 8th 2020, 603 adverse events (AEs) were reported in the entire cohort; 313 (52%) were related to study drug and 127 (21%) were serious adverse events. 286 (47%) occurred in the induction treatment (see table 1). Of these, 69 (24%) were CTC grade 3 and 18 (6%) CTC grade 4, 4 had a fatal outcome. Most common AEs in the induction were infusion-related reactions, neutropenia, thrombocytopenia, anemia, nasopharyngitis, headache, and fatigue [Table 2].
Summary/Conclusion:
Sequential treatment with Ben debulking, followed by Obi and Ide induction and maintenance achieved responses and even undetectable MRD levels in CLL patients with high-risk disease and extensive prior therapy. However, the study also confirmed the known toxicities of Ide. In light of the current, alternative therapeutic options, the BCG regimen reported here should be used with caution, but represents an alternative treatment option if ibrutinib and venetoclax have failed.
Cramer:Gilead: Other: travel support, Research Funding; F. Hoffmann-LaRoche: Honoraria, Other: travel support, Research Funding; Acerta: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; AstraZeneca: Consultancy, Honoraria, Other: travel support, Research Funding; Beigene: Research Funding; AbbVie: Honoraria, Other: travel support; Novartis: Consultancy, Research Funding; Janssen-Cilag: Consultancy, Honoraria, Other: travel support, Research Funding. Von Tresckow:Celgene: Other: travel grants; AbbVie: Honoraria; Janssen-Cilag: Honoraria, Other: travel grants, Research Funding; F. Hoffmann-LaRoche: Honoraria, Other: travel grants, Research Funding. Fink:AbbVie: Other: travel grants; Janssen: Honoraria; Celgene: Research Funding. Tausch:Janssen-Cilag: Consultancy, Honoraria, Research Funding; Roche: Consultancy, Honoraria, Research Funding; AbbVie: Consultancy, Honoraria, Research Funding. Knauf:Janssen-Cilag: Honoraria; AbbVie: Consultancy, Honoraria; AMGEN: Consultancy, Honoraria; BMS: Consultancy, Honoraria; Celgene: Consultancy, Honoraria; Gilead: Consultancy, Honoraria; Mundipharma: Honoraria. Al-Sawaf:BeiGene: Research Funding; AbbVie: Consultancy, Honoraria, Other: personal fees, Research Funding; Roche: Consultancy, Honoraria, Other: personal fees, Research Funding; Gilead: Consultancy, Honoraria, Other: personal fees; Janssen: Consultancy, Honoraria, Other: personal fees, Research Funding; AstraZeneca: Consultancy, Honoraria, Other: personal fees. Langerbeins:AbbVie: Honoraria, Other: travel grants, Research Funding; F. Hoffmann-LaRoche: Honoraria, Other: travel grants, Research Funding; Janssen-Cilag: Honoraria, Other: travel grants, Research Funding; Mundipharma: Honoraria, Other: travel grants, Research Funding. Fischer:F. Hoffmann-La Roche: Honoraria, Other: travel grants; AbbVie: Honoraria. Kreuzer:Hoffmann-La Roche: Consultancy, Honoraria, Research Funding; AbbVie: Consultancy, Honoraria, Research Funding; Amgen: Consultancy, Honoraria, Research Funding; Gilead: Consultancy, Honoraria, Research Funding; Janssen-Cilag: Consultancy, Honoraria, Research Funding; Mundipharma: Consultancy, Honoraria, Research Funding. Ritgen:Gilead: Other: travel grants; BMS: Consultancy, Honoraria, Other: travel grants; F. Hoffman-La Roche: Consultancy, Honoraria, Other: travel grants, Research Funding; Pfizer: Consultancy, Honoraria. Kneba:AbbVie: Consultancy, Honoraria, Other: travel support, Research Funding; F. Hoffmann-LaRoche: Consultancy, Honoraria, Other: travel support, Research Funding; Gilead: Consultancy, Honoraria, Other: travel support, Research Funding; Janssen-Cilag: Consultancy, Honoraria, Other: travel support, Research Funding; Mundipharma: Consultancy, Honoraria, Other: travel support, Research Funding. Wendtner:Mundipharma: Consultancy, Honoraria, Other: travel support, Research Funding; Pharmacyclics: Consultancy, Honoraria, Other: travel support, Research Funding; AbbVie: Consultancy, Honoraria, Other: travel support, Research Funding; Gilead: Consultancy, Honoraria, Other: travel support, Research Funding; Hoffmann-LaRoche: Consultancy, Honoraria, Other: travel support, Research Funding; Genentech: Consultancy, Honoraria, Other: travel support, Research Funding; Janssen-Cilag: Consultancy, Honoraria, Other: travel support, Research Funding; GlaxoSmithKline: Consultancy, Honoraria, Other: travel support, Research Funding. Stilgenbauer:Pharmacyclics: Consultancy, Honoraria, Other, Research Funding; Novartis: Consultancy, Honoraria, Other, Research Funding; Mundipharma: Consultancy, Honoraria, Other, Research Funding; Janssen-Cilag: Consultancy, Honoraria, Other: travel support, Research Funding; GlaxoSmithKline: Consultancy, Honoraria, Other: travel support, Research Funding; Gilead: Consultancy, Honoraria, Other: travel support, Research Funding; Genzyme: Consultancy, Honoraria, Other: travel support, Research Funding; Genentech: Consultancy, Honoraria, Other: travel support, Research Funding; F. Hoffmann-LaRoche: Consultancy, Honoraria, Other: travel support, Research Funding; Celgene: Consultancy, Honoraria, Other: travel support, Research Funding; Boehringer-Ingelheim: Consultancy, Honoraria, Other: travel support, Research Funding; Amgen: Consultancy, Honoraria, Other: travel support, Research Funding; AbbVie: Consultancy, Honoraria, Other: travel support, Research Funding. Eichhorst:Janssen-Cilag: Consultancy, Honoraria, Other: travel support, Research Funding; Celgene: Consultancy, Honoraria, Other: travel support, Research Funding; Novartis: Consultancy, Honoraria, Other: travel support, Research Funding; ArQule: Consultancy, Honoraria, Other: travel support, Research Funding; BeiGene: Consultancy, Honoraria, Other: travel support, Research Funding; Gilead: Consultancy, Honoraria, Other: travel support, Research Funding; AstraZeneca: Consultancy, Honoraria, Other: travel support, Research Funding; Oxford Biomedica: Consultancy, Honoraria, Other: travel support, Research Funding; F. Hoffmann-LaRoche: Consultancy, Honoraria, Other: travel support, Research Funding; AbbVie: Consultancy, Honoraria, Other: travel support, Research Funding. Hallek:F. Hoffmann-LaRoche: Consultancy, Honoraria, Research Funding; Gilead: Consultancy, Honoraria, Research Funding; Mundipharma: Consultancy, Honoraria, Research Funding; Janssen: Consultancy, Honoraria, Research Funding; Celgene: Consultancy, Honoraria, Research Funding; AbbVie: Consultancy, Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.